Mini Caps
P3 level 24/7 pathogen testing
Epidemic outbreaks of Bird flu Influenza, Zika and recently Covid-19 have made it clear we need fast and flexible research capacity to develop assays for new pathogenic agents and screen for molecular targets with therapeutic activity.
MiniCaps automated pathogen assay platform

Nucomat and MBraun have engineered a compact and flexible automated platform for 24/7 cell culture based pathogen assays and screening inside a top notch sealed enclosure at the highest standards of biosafety. Building on the experience from the larger Caps-IT platform for automated P3+ pathogen screening, at the Rega Institute for infectious diseases at KULEUVEN (Belgium), the lab staff can operate a P3+ process from a more convenient P2 work environment to work with new or poorly characterized pathogens.
Your benefits
- Operator safety and comfort, environment safety.
- Traceable and repeatable hands-off 24/7 operation.
- Compact solution; reduced cost to a complete P3 lab.
MiniCaps Features
- 2 autonomous automated microplate cell incubators: 120 microtiter plates each
- Standard ANSI-SLAS 96 and 384 well format
- maintain and process cell cultures at 30 ºC to 42ºC, 5% CO2, humidity.
- Sealed back window for safe visual inspection.
- Autonomous disinfection cycles.
- Liquid handler station with a configurable 1m deck.
- Microplate reagent dispenser
- 96/384
- plate delidder
- Plate washer
- 96/384
- buffer exchange
- ELISA assays.
- Multimode plate reader
- 96/384
- colorimetry
- fluorescence
- luminescence.
- High Content Imager: imaging and analysis in microtiter plates
- epifluorescence
- confocal option
- custom protocols in imager software
- stained and live cell assays.
- Safety lock: decontamination to import and export materials in closed ISO cages.
- Space saving overhead Cartesian robotics
- Reproducible operation
- Glove Box access for manual actions.
Central web-based software
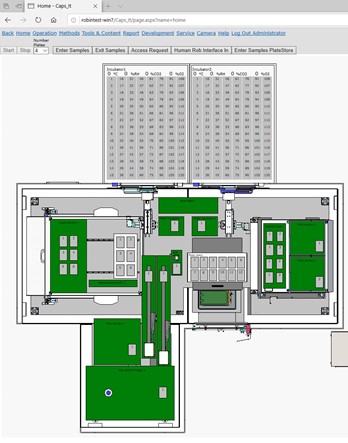
- ROBIN scheduler, Nucomat’s proprietary real-time event-driven scheduling software
- priority process scheduling.
- Web interface:
- run samples
- oversee the processes
- edit protocols
- Error handling, smart retry, run recovery.
- Sample Database
- Barcode ID sample tracking from registration to result
- event logging
- data import, export and reporting, LIMS compatible.
- Offline reader data handling
- database result links.
- Remote operation
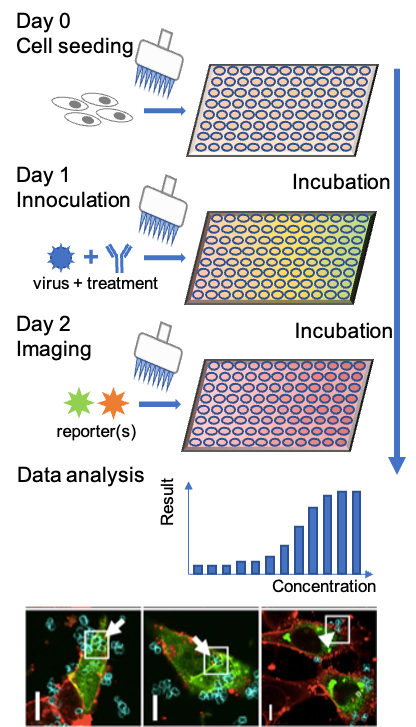
Assay procedures and platform flexibility
- Flexible multi-assay operation
- Example protocols for Seeding, incubation, harvesting, plate replication, reading, HCI monitoring, and end-point testing.
- Edit or build your own protocols.
- Run multiple protocols in parallel.
- Switch plates from one protocol to another.
- Replicate plates for multiple readouts.
- Maintain cell culture while decontaminating platform.